 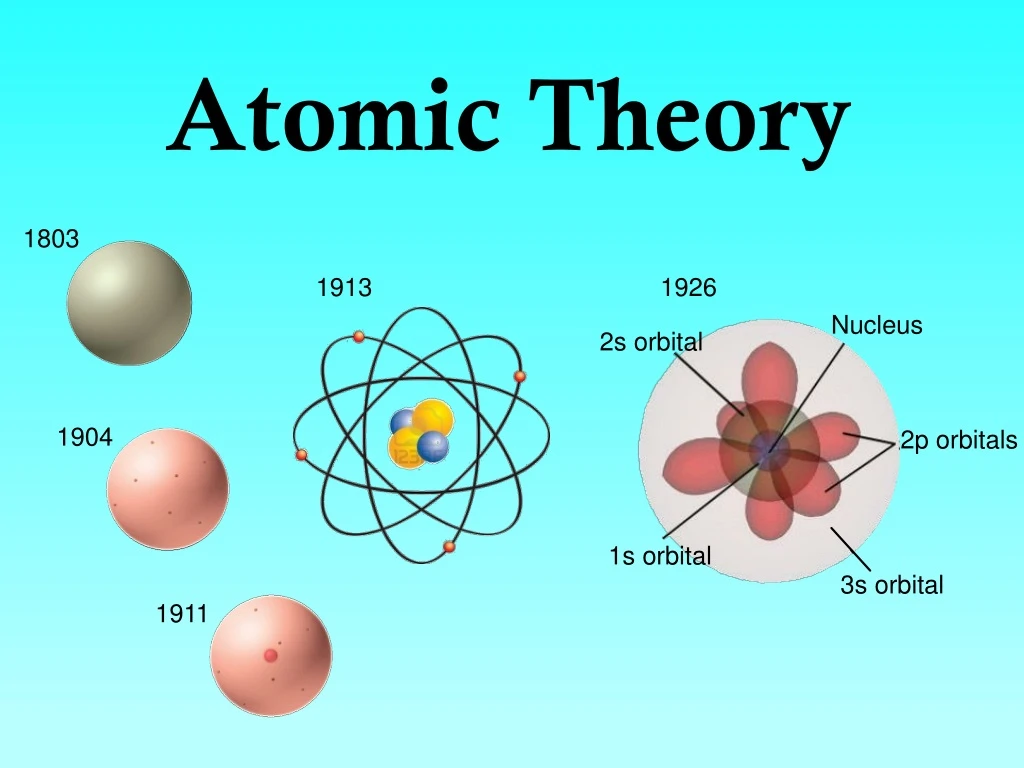
It provided a framework for understanding the behavior of matter at the atomic level, paving the way for further discoveries and developments in the field. His atomic theory served as the basis for subsequent advancements in atomic and molecular physics. However, Dalton’s ideas gradually gained acceptance and eventually became the cornerstone of modern atomic theory.ĭalton’s legacy in the scientific community is profound. Some objected to his atomic theory, challenging the idea that atoms are indivisible and uniform in nature. His research ultimately led to the understanding that color blindness is a result of defects in the perception of specific colors.ĭespite his significant contributions, Dalton faced criticism and skepticism from some scientists of his time. He conducted experiments on his own vision and the vision of others, meticulously documenting his findings. This observation led him to propose the law of multiple proportions, a fundamental principle in stoichiometry.Īdditionally, Dalton’s meticulous study of color blindness further showcased his scientific acumen. He analyzed the proportions in which elements combine to form compounds and observed that elements can combine in different ratios to form multiple compounds. This observation, known as Dalton’s law of partial pressures, provided valuable insights into the properties of gases and contributed to the development of the kinetic theory of gases.ĭalton’s investigations into chemical reactions and the composition of compounds also played a significant role in the development of his atomic theory. He observed that the total pressure exerted by a mixture of gases is equal to the sum of the pressures that each gas would exert if it occupied the same volume alone. One of Dalton’s notable experiments involved investigating the behavior of gases. Throughout his career, he conducted numerous experiments to support and refine his ideas. It provided a coherent framework for understanding chemical reactions and the behavior of matter, establishing the foundation for modern chemistry.ĭalton’s atomic theory was not merely a product of speculation but was grounded in extensive experimental work. Dalton also introduced the concept of atomic weights, allowing chemists to determine the ratios in which different elements combine to form compounds.ĭalton’s atomic theory represented a significant departure from prevailing ideas, such as the phlogiston theory and the four-element theory. Furthermore, he suggested that each element is made up of atoms that are identical in size, mass, and chemical properties. He proposed that atoms are indestructible and retain their identity during chemical reactions. In 1803, he published “A New System of Chemical Philosophy,” a groundbreaking work that outlined his revolutionary ideas on the nature of matter.Īccording to Dalton’s atomic theory, matter is composed of tiny, indivisible particles called atoms. Dalton continued to pursue his studies while teaching, deepening his knowledge of mathematics, chemistry, and physics.ĭalton’s most significant contribution to science was his formulation of the atomic theory. His talent and passion for these subjects caught the attention of local scholars, leading to his appointment as a teacher at the age of 12. From a young age, Dalton showed a keen interest in the natural world and had a particular fascination with meteorology.ĭalton’s formal education began at the Quaker school in Eaglesfield, where he excelled in mathematics and science. He was the son of a weaver and grew up in a modest, hardworking family.  
John Dalton was born on September 6, 1766, in Eaglesfield, Cumberland, England. This comprehensive article explores Dalton’s life, his contributions to atomic theory, his groundbreaking experiments, and his lasting legacy in the scientific community. His pioneering work in the late 18th and early 19th centuries laid the foundation for our understanding of the atom and revolutionized the field of chemistry. John Dalton, a renowned English chemist and physicist, is widely recognized as the father of modern atomic theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
